Case1.Recurrence after surgery for cancer of the upper jaw
Name:Recurrence after surgery for cancer of the upper jaw
Stage: ⅣB
Age/Sex:70-year-old male
Symptoms&Progress
The patient had recurrence of cancer after the surgery and was told in the year X that he would live only for another 6 months, and all treatments were stopped. Sincethen, the patient received no anticancer treatment orradiotherapy, and volunteered to have Gerson’s diet
therapy along with yoga therapy and hot spring therapy.
After that, he also had high dose vitamin C drips once or twice a week. The progression of the tumor became veryslow, and the patient visited our hospital for the first time in the year X + 3.
However, the tumor continued to advance, and the patient started taking X6 1P/day concurrently from the year X+3.
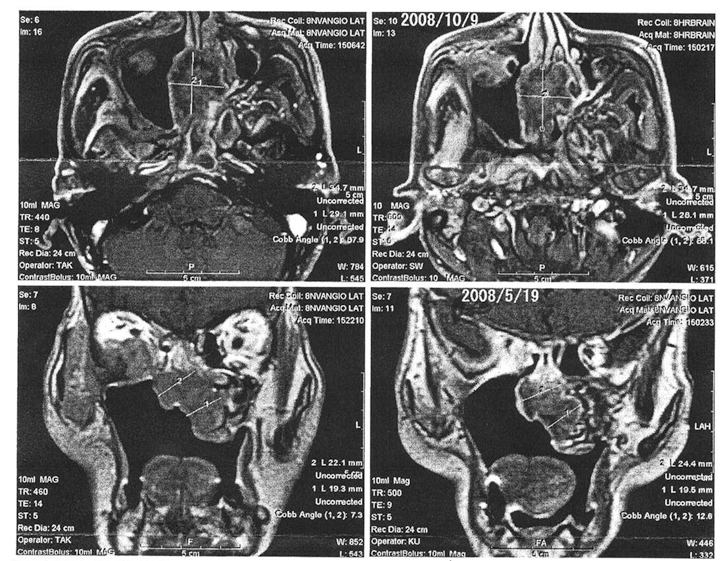
Three months from the start of the X6 treatment, MRI showed that the tumor had not increased in size form the previous observation, and thus the cessation of tumor enlargement could be confirmed for the first time through imaging.
After that, X6 was continued at the increased dose of 3P/day, and concurrently, the diet therapy and high concentration vitamin C drip once or twice a week were also continued.
After one month on X6 3P/day, MRI showed shrinkage of the tumor for the first time.
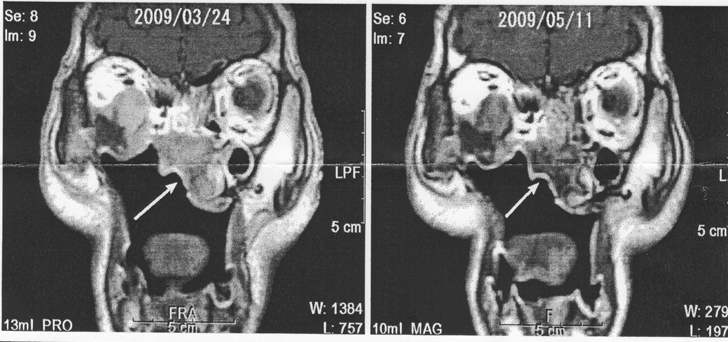
The MRI finding after two months on X6 3P/day led to the comment, “Staining of the tumor by gadolinium has clearly decreased, which suggested decreased tumor activity or decreased blood flow, intravenous injection of gadolinium was smooth, and there was no leakage of the contrast agent”.
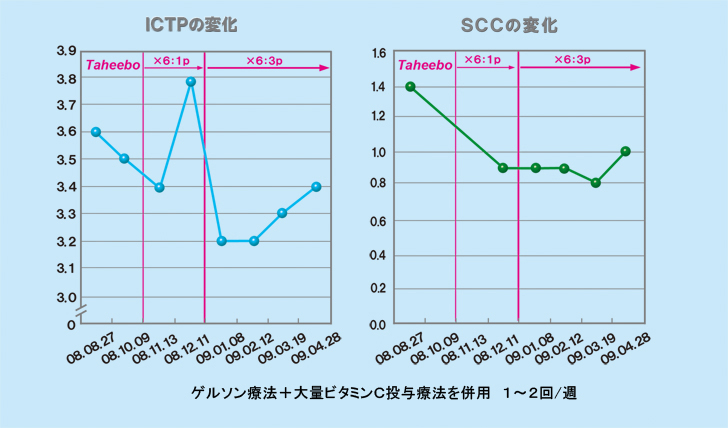
During this period, the tumor marker ICTP increased once and then decreased sharply, whereas SCC decreased continuously.
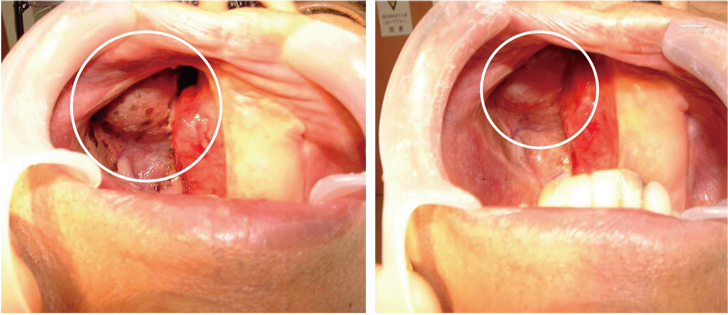
Intraoral observations also showed that the tumor that had been advancing slowly had shrunk (Fig. 5 Top) three months after the dose of X6 was increased form 1P to 3P/day.
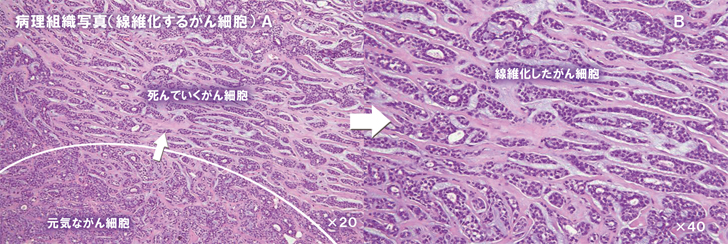
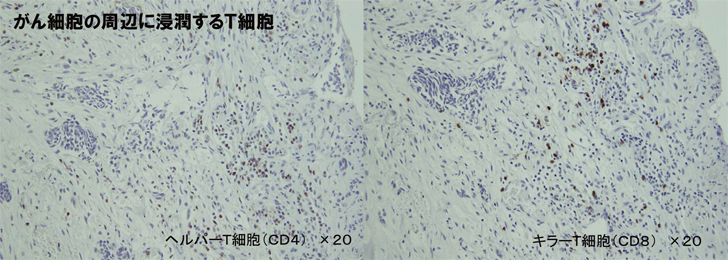
Conclusions from Case 3
The tumor markers ICTP and SCC decreased after the X6 treatment started and clinically also the tumor was seen to have shrunk, for the first time. After three months on X6 3P/day, the MRI showed a clear decrease in uptake of the contrast agent, which suggested lowered activity of the tumor.
The X6 3P/day treatment changed into parasympathetic dominance favorably (decreased the G/L
ratio) (data not shown). Besides this, the patient showed no biochemical or clinical abnormality during the X6 treatment.
The results of this case also suggested dose dependence of the effect of NQ801 and its safety.
The patient continues to be on X6 3P/day concurrently with diet therapy and high dose vitamin C drip, and the tumor is shrinking and has reached about half its former size.
Areas for future studies
Clinical investigations of the present study have suggested that the anticancer effect of NQ801 is dose dependent, and confirmed the safety of NQ801 up to 5.4 mg/day. In the future, it is necessary to investigate the dose requirement of NQ801, when used in combination with anticancer drugs in specific cases, by using it on a larger number of patients in integrative treatment of cancer.
Literature
Case2. Both inoperable pancreatic cancer and colon cancer
Japanese Jounal of Complementary and Alternative Medicine (Vol.8 No.2 2011)
Yoshio TAKEDA (Takeda Internal and Gastrointestinal Clinic)
【ABSTRACT】
A 58 year-old man affected by both inoperable pancreatic cancer and colon cancer was treated withchemotherapy gemcitabine and TS1. FRx6 which contains six-fold effective ingredient NQ801, was also given simultaneously. Partial response of both pancreatic and colon cancer was obtained by chemotherapy and FRx6.
Further examination of combined therapy will be needed.
1.Introduction
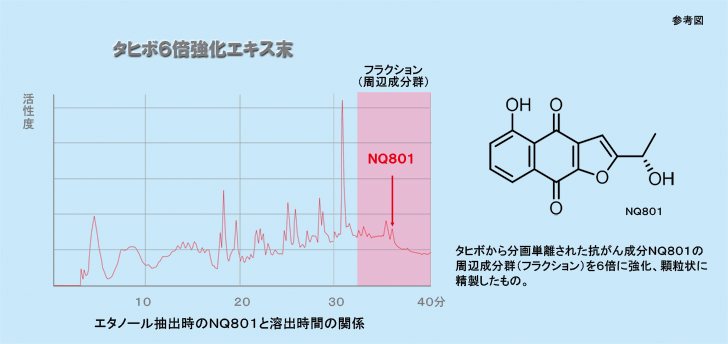
“Taheebo”is hot-Water extract taken from the bark of the Tabebuia avellanedae tree of the Bignoniaceae family found in Brazil of South America and it is widely used as a folk remedy for variety diseases in Brazil. Ueda, et al, reported that naphthoquinone, a Taheebo tea extract, inhibited the activation of the initial expression of TPA -induced EB virus and it also inhibited tumor promoter activity in vitro. Its physiologically powerful and effective ingredient is 5-hydroxy-2-(1-hydroxy-ethyl)-naphtho [2, 3b] fur an-4,9-dione and its code name is NQ801. This effective ingredient is extracted from the Tabebuia avellanedae (Taheebo) trees those are native to a specific area Regression of double cancer was observed in a patient affected by both inoperable pancreatic cancer and colon cancer after Taheebo extract FRx6, in which the concentration of NQ801 was multiplied by six, was simultaneously administered with gemcitabine and TSI chemotherapy. Therefore, this case is reported.
2.Methods
A 58-year-old man was diagnosed as both inoperable Stage Ⅳb pancreatic head cancer and advanced ascending colon cancer which were double primary cancer at a certain hospital on November 10, 2008.
Treatment was started from the end of November 2008, with gemcitabine administration twice in two weeks, however, no gemcitabine was given in the following one week.
Four capsules of TS1 were also given per os everyday for two weeks simultaneously, however, no TS1 was given for the following one week. This treatment was repeated every three weeks.
On November 28, 2008, CA19姐9tumor marker level was 895 μ/mL. There was an irregular low echogenic tumor in the pancreatic head, which corresponded to pancreatic cancer of 32.4×35.7 mm in size.
From December 14, 2008 to June 23, 2009, the patient obtained Taheebo extract FRx6 from a company and he took it per os.
On December 27, 2008, the CA19-9 tumor marker level was 42 μ/mL.
On January 24, 2009, the size of the pancreatic head cancer was 30.4×21.5 mm.
On February 28, 2009, the size of the pancreatic head cancer was 15.8×13.4 mm.
On March 10, 2009, the CA19-9 tumor marker level normalized to 4.9 μ/mL.
On March 28, 2009, the size of the pancreatic bead cancer was 13.5×10.5 mm.
On May 12, 2009, the CA19-9 tumor marker level was 4.4 μ/mL.
On May 30, 2009, the pancreatic cancer could not be detected in an abdominal ultrasound sonography.
In June 2009, the pancreatic head cancer was not almost seen with CT scan test either, and partial response (PR), very close to complete response (CR), was observed. The advanced colon cancer was also markedly reduced to a tiny scar lesion and it was also evaluated as PR.
3. Results
One case of a double primary cancer patient affected by both inoperable pancreatic head cancer and advanced colon cancer resulted in tumor regression as PR after FRx6 was simultaneously administered during gemcitabine and TSI chemotherapy.
4. Discussion
Taheebo extract from Tabebuia avellanedae contained the active ingredient “NQ801”and displayed direct action on cancer cells such as selective toxicity, apoptosis induction, angiogenesis inhibition, and inhibition of both metastasis and invasion potential. It also had
immunostimulatory effect as indirect action. Complete regression in a patient with end stage hepatocellular carcinoma and liver cirrhosis has already reported after ingestion of Taheebo tea6. According to Sudo7 et al, in a Phase II trial on advanced pancreatic cancer by using both gemcitabine and TS1 in 21 subjects resulted in PR assessment for 2 patients (9.5%), stable disease (RECIST: Response Evaluation Criteria In Solid Tumors) for 9 patients (43%), with CA19-9level decreasing at least 50% in 5 of 18 subjects.
When oxaliplatin and gemcitabine were concurrently administered to 43 colon cancer patients, one case resulted in PRS. According to Taiho Pharmaceutical, in a Phase II trial of colon cancer using TS1, while there were no CR results, 42 subjects out of 129 (32.6%) were evaluated as PR. On the other hand, it was reported that, when β-Lapachone, one substance of naphthoquinone compaound, a NQ801-like compound, was used concurrently with taxol,β-Lapachone had a synergistic anti-tumor effect and no side effects were observed in the mice themselves9.
It can be thought that the tumor regression in the double primary cancer of inoperable pancreatic cancer and advanced colon cancer experienced this time was due to effect of both the gemcitabine and TS1 mainly, however, the synergistic effect of FRx6 cannot be excluded. Moreover, the fact that the anti-tumor effect of β-Lapachone and taxol is synergistic9 suggests the possibility of a synergistic effect with chemotherapy and FRx6. NQ801 is a substance discovered by Japanese scientists and it is expected that various clinical trials will be conducted in the future.
Literature
1) Walter Radames Accorsi. Miracles of Taheebo extract. Kobe Shimbun Publishing Center. 1988; 48.
2) Ueda S, Umemura T, Dohguichi K, et al. Production of anti-tumor promoting furanonaphthoquinones in Tabebuia Avellanedae cell culture. Phytochemistry 1994; 36: 323-325.
3) Hirata S. Clinical examination of NQ801 extracted from Taheebo in integrative medicine for cancer. International journal of Integrative Medicine 2010 ; 2 (1) : 119-127.
4) Ebina T. Anti”tumor effect of hot water extract of Taheebo, Tabebuia avellanedae grown in South America. Biotherapy 1998 ;12 : 495”500.
5) Ebina T. Anti-tumor effect of hot water extract of Taheebo comparing with other biological substances. Biotherapy 2002; 61: 321-327.
6) Takeda Y, Togashi H, Shinzawa H, et al. Spontaneous regression of hepatocellular carcinoma and review of literature. J Gastroenterol Hepatol. 2000; 15(9): 965-966.
7) Sudo K, Yamaguchi T, Nakamura K, et al. Phase II study of S-1 in patients with gemcitabine-resistant advanced pancreatic cancer. Cancer Chemother Pharmacol. 2011; 67 (2): 249-254.
8) Shibata S, Chow W, Frankel P, et al. A phase I study of oxaliplatin in combination with gemcitabine: correlation of clinical outcome with gene expression. Cancer Chemother Pharmacol. 2007; 59(4):549-557.
9) Pardee AB, Li YZ, Li CJ, et al. Cancer therapy withbeta-lapachone. Curr Cancer Drug Targets. 2002; 2(3): 227-242.



 〒541-0054
Ikeman Building 8F, 2-2-5 Minamihonmachi, Chuo-ku, Osaka-shi, Osaka JAPAN
〒541-0054
Ikeman Building 8F, 2-2-5 Minamihonmachi, Chuo-ku, Osaka-shi, Osaka JAPAN